Build the Science
Establish the biomarker, validate the mechanism, build the patent estate.
VolitionRx (NYSE American: VNRX) could be early on a similar path Exact Sciences (NASDAQ: EXAS), Veracyte (NASDAQ: VCYT), and Castle Biosciences (NASDAQ: CSTL) walked from launch to scale — three publicly-traded cancer diagnostics platforms. Same DNA. Different stage.
Exact Sciences, Veracyte, and Castle Biosciences didn’t start as giants. They started as platforms — a focused biomarker, a first commercial wedge, distribution earned not assumed. Then a slow-then-fast climb into a multi-billion-dollar category. Four stages. Three companies. One repeating pattern.
Most retail investors miss the early-platform stage where each name traded at a fraction of today’s value. By the time the pattern is obvious in the price, the asymmetric part is gone.
Establish the biomarker, validate the mechanism, build the patent estate.
Run the clinical cohort. Print specificity and sensitivity that move the literature.
License or distribute through partners with the doors and the reimbursement.
Compound a multi-billion-dollar category as adoption begins to flywheel.
Where Exact · Veracyte · Castle spent years
Three points on the same arc. Exact Sciences at full scale, Veracyte through the proven decade, Castle Biosciences mid-journey. Each one started at a stage similar to where VolitionRx sits today — a single validated test, an early commercial wedge, a reimbursement pathway in motion. Same DNA. Different stages of the same shape.
Cologuard launched in Q4 2014 with roughly 4,000 tests and $1.5M in revenue. By 2024, Exact reported $2.76 billion in total revenue. Single test, validated, reimbursed, scaled. VolitionRx received its first clinical certification sale of Nu.Q® Cancer at Hospices Civils de Lyon in November 2025 — a comparable starting point, eleven years offset.
Veracyte launched the Afirma thyroid test in 2011 at $2.65M in revenue. By 2020, organic growth pushed it to $117M — a 44× lift on a single test in nine years. They then leveraged that organic success to springboard major M&A deals (Decipher and HalioDx, both 2021), pushing revenue to roughly $517M and market cap to ~$2.7B. Single test → platform.
Castle launched DecisionDx-Melanoma in March 2013. Revenue scaled from $13.75M (2017) to $344M (2025) — 25× in eight years on a single specific molecular test. Now ~$725M market cap. The most direct mid-journey reference for what the next five years could look like for VolitionRx.
VolitionRx remains an earlier-stage diagnostics company. Comparisons to Exact Sciences, Veracyte, and Castle Biosciences are for industry-context purposes only and do not imply equivalent regulatory status, revenue scale, market adoption, or future valuation.
Logos are easy to dismiss. Revenue trajectories aren’t.
Look at where each one started. Exact Sciences ran roughly 4,000 Cologuard tests in Q4 2014 — about $1.5 million in revenue at launch. By 2024, Exact reported $2.76 billion in total revenue. Veracyte launched its Afirma thyroid test in 2011 at $2.65 million in revenue; by 2020, organic growth had pushed it to $117M — a 44× lift on a single test before the company began acquiring complementary platforms (Decipher, HalioDx) that took 2025 revenue to ~$517M. Castle Biosciences launched DecisionDx-Melanoma in 2013; revenue scaled from $13.75M (2017) to $344M (2025).
Now anchor those starts against where Volition sits today. FY2025 revenue: $1.7 million, +40% YoY, with Q4 2025 up 133% YoY. That’s the same revenue order of magnitude where Exact stood when Cologuard first hit the market and where Veracyte stood when Afirma launched. Eleven years offset on the same arc.
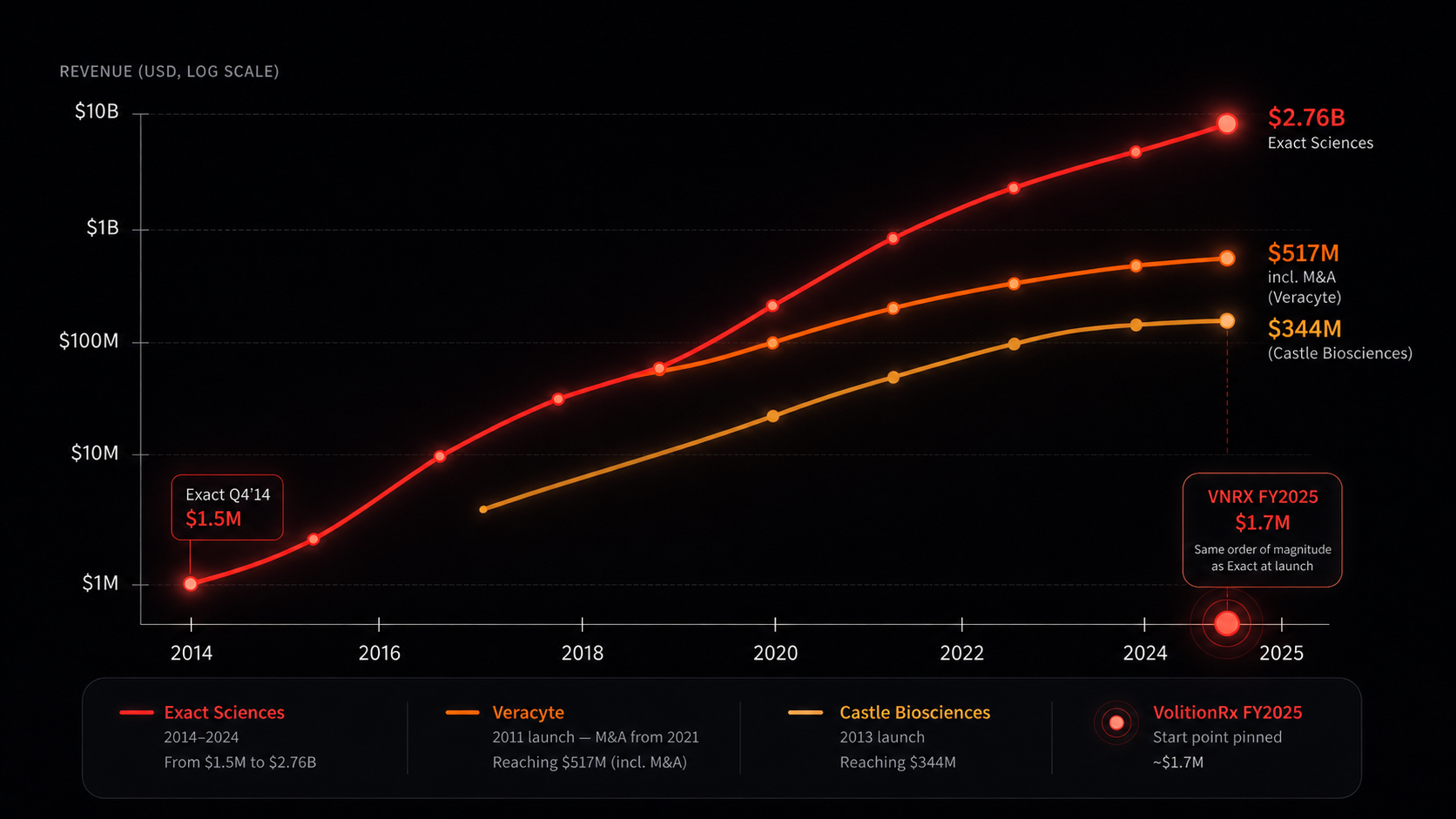
Diagnostic companies often look small before clinical validation, reimbursement, and distribution begin to compound.
Stripped of the jargon, the Nu.Q® platform does something simple to describe and hard to do.
Your blood carries millions of tiny spools called nucleosomes — the structures that hold DNA inside every cell. As cells live, divide, and die, they release nucleosomes into circulation, and different cell types leave different fingerprints. Cancer cells leave one fingerprint. Sepsis-driven immune cells leave another. Stressed organs and damaged tissue leave their own. Volition’s Nu.Q® platform is designed to count those fingerprints from a single blood draw and read them.
One sample. One workflow. A panel of potential answers — cancer signal, sepsis signal, immune-response signal, veterinary cancer signal, and the early outline of a finger-prick point-of-care signal that the company demonstrated for the first time on April 29, 2026.
The science isn’t a slogan; it’s a counting problem the company has spent 15 years learning how to solve. That’s the foundation everything else on this page is built on.

Cells live, divide, and die. As they die, they release nucleosomes into the bloodstream — constantly, in everyone, all the time.
Cancer cells, infected cells, immune cells, and damaged cells each leave a different chemical signature on the nucleosomes they shed.
Nu.Q® counts those signatures from a single blood sample — the same kind a physician runs for cholesterol.
The platform lesson — one platform, many verticals — is the easiest to claim and the hardest to prove. Below is what Volition has actually moved into the field, with a date and a partner attached to each branch.
Five branches off one core science. Each branch has a date and a partner. That’s what platform expansion looks like in the field, not on a slide.
Most biotech investors auto-discount the veterinary line. They shouldn’t, because the vet business is doing the de-risking work — proving the platform is licensable, milestone-able, and globally distributable today, not in some future scenario.
$10M upfront + $13M achieved milestones · up to $5M pending feline lymphoma trigger
VolitionRx has received $23 million in upfront and milestone payments under its exclusive agreement with Heska, an Antech company, for in-hospital access to the Nu.Q® Vet Cancer Test. An additional $5 million milestone remains pending — tied to a feline lymphoma commercialization or peer-reviewed publication trigger. IDEXX launched the IDEXX Nu.Q® Canine Cancer Test in January 2023.
20+ Countries
Antech/Heska · IDEXX · Fujifilm Vet Japan (1,700+ Hospitals) · Vita Genomics · DNA Tech · Nationwide · VPG
Multi-Cancer Early Detection (MCED) + Minimal Residual Disease (MRD) · company estimate
If a veterinary cancer testing agreement can generate $23 million in upfront and milestone payments, the potential scale of human diagnostics — where the addressable markets in cancer, sepsis, and disease monitoring are significantly larger — deserves a closer look.
Active Partner Footprint
Werfen · Hologic · Mayo Clinic · Hospices Civils de Lyon
Veterinary success does not guarantee human commercial success, but it gives investors a real-world signal: Volition’s platform has already attracted commercial partners and milestone economics.
The hardest problem in early cancer detection has always been the same: cancer DNA hides in blood at a vanishingly low ratio. Find it reliably without false positives, and you’ve cleared the gate that keeps most multi-cancer screening tests stuck in research mode.
In its blinded Capture-Seq™ validation cohort, Volition reported 95% sensitivity for Stage I and II cancers with 95% specificity, on top of an enrichment step that delivers 180-fold concentration of cancer DNA at 99% purity. The cohort: 81 subjects — 59 cancer patients (lung and colorectal) and 22 healthy controls. Across all stages, sensitivity ran 93%.
Volition has stated Capture-Seq™ targets a $36 billion TAM spanning multi-cancer early detection (MCED) and minimal residual disease (MRD) — the same diagnostic categories where Abbott just paid $23 billion for one franchise and Roche just paid up to $595 million for another.
Enrichment of cancer DNA from blood
Purity in the cancer-DNA isolation step
Sensitivity for Stage I and II cancers
Specificity across the validation cohort
Subjects · 59 cancer (lung + colorectal) + 22 healthy controls
TAM — MCED + MRD (company estimate)
We believe the ‘needle in a haystack’ problem that has held back multi-cancer early detection has now been solved.
Jake Micallef, CSO, VolitionRx · December 2025 Capture-Seq announcement
The cohort is small, peer-reviewed publication is pending, and reimbursement remains a multi-year process. None of that changes the fact that the data prints in the same tier of sensitivity and specificity that the comparable giants used to compound.
The platform isn’t done expanding. On April 29, 2026, Volition announced successful finger-prick detection of nucleosomes in capillary blood from critically ill sepsis patients, using a lateral flow prototype — the same product class behind the home COVID-19 tests that became familiar overnight.
The company described the result as a potential path toward scalable point-of-care testing — ER bedside, ICU, doctor’s office, or potentially home self-test — for one of medicine’s most urgent decisions: is this patient septic, and how fast does treatment need to start?
It’s a prototype. It needs validation, regulatory clearance, and distribution before it touches a real patient outside research. But it answers the question the radial diagram raises in §6 — is the point-of-care branch real? — with a date and a result.
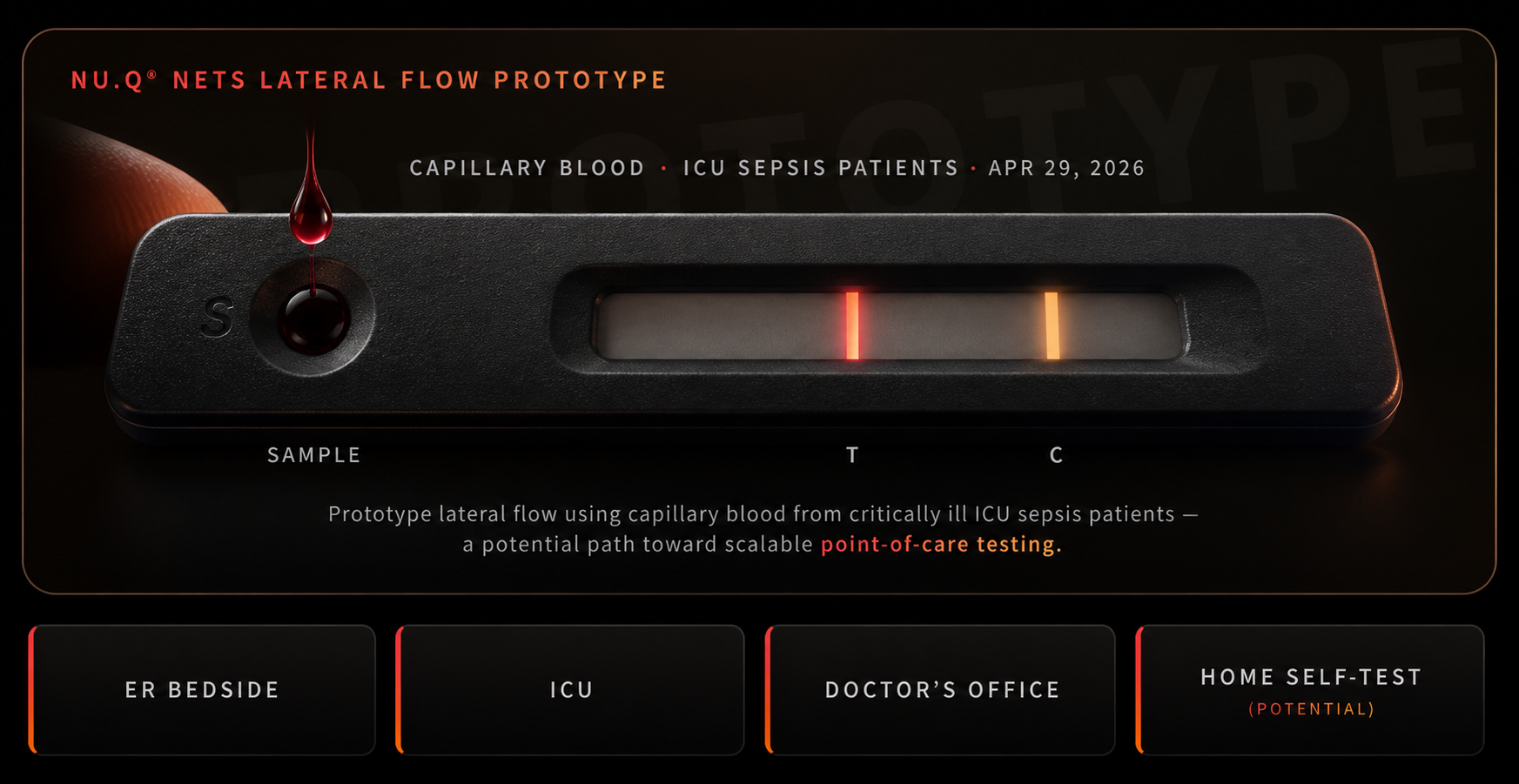
Conceptual illustration. Lateral flow validation announced April 29, 2026 in critically ill ICU sepsis patients; production-grade cassette hardware is not yet finalized and the actual prototype may differ in form factor.
Regulatory clearance secured for hospital labs across the European Union.
Selected as the only biomarker in the French government sepsis program.
Active hospital evaluators as of December 31, 2025.
First revenue from CE-marked automated product recorded.
Revvity reimbursement collaboration in preparation for 2026.
Capillary blood detection demonstrated in critically ill ICU sepsis patients.
The comparable framework lives or dies on real, recent transaction prices. Here’s what the major acquirers have actually written checks for in the last 24 months. Set those numbers against where Volition trades today.
MRD, pre-commercial liquid biopsy.
Proteomics platform, closed.
Cancer screening / MCED. Closed at $105/share cash.
MRD, announced.
The platform Volition is building targets the same categories — MCED and MRD — and Volition has publicly stated it is in active discussions with roughly ten leading diagnostic and liquid-biopsy companies about potential licensing arrangements (company update, February 2026). A licensing deal isn’t a takeout, and “active discussions” aren’t signed contracts.
But the major acquirers aren’t watching from the sidelines anymore. They’re writing checks.
For industry-context purposes only — comparisons do not imply equivalent regulatory status, scale, or future valuation.
This isn’t a “company has always had a tight structure” story. On April 28, 2026, VolitionRx executed a 1-for-20 reverse split — taking the share count from approximately 131 million down to roughly 8 million shares outstanding. Public float now sits near 4.7 million. The reset is brand new.
What the cleaner share count does is change the math: catalysts move the price differently when there are far fewer shares for them to push against. Structural math, not narrative. Roughly 3×–23× tighter than the comparable peer set — the tightest float in the comp framework.
A capital structure reset — a cleaner share count landing in the same window as a stack of commercialization catalysts.
The biggest diagnostics winners often share the same DNA: Validated science. Commercial pathway. Scalable testing category. Large unmet market. Exact Sciences scaled non-invasive cancer screening into billions in annual revenue. Veracyte built a single thyroid test (Afirma) into a multi-vertical diagnostics platform. Castle Biosciences scaled DecisionDx-Melanoma into a $344M molecular dermatology business. Different molecules, different starting wedges, the same four-pillar shape underneath.
95% Stage I–II sensitivity (Capture-Seq, 81-subject cohort)
$23M received (Antech/Heska), 20+ countries
5 platform branches, dated partners on each
$36B MCED + MRD (company estimate)
VolitionRx is earlier-stage, but it is building around the same investor-relevant ingredients: A proprietary testing platform. Multiple disease applications. Commercial veterinary traction. Human diagnostic catalysts. Partner-driven distribution.
The science is reading at the levels that move giants — 95% Stage I–II sensitivity, 95% specificity, 180-fold enrichment, 99% purity. The commercial pathway is already cash-positive on the veterinary side at $23 million received. The category is scalable — five branches off one core, each with a date and a partner attached. And the unmet markets are the largest in modern medicine: $36 billion in MCED and MRD alone, plus sepsis, plus immune response, plus the long tail of point-of-care possibilities the April 29 prototype just opened.
That’s why VNRX deserves attention now, before the market decides whether Nu.Q® can move from promising platform to scaled diagnostics company.
Same DNA. Different stage. The shape is already on the page.
The catalyst calendar runs through 2026. We send updates only on the material moments — no weekly newsletters, no marketing churn.
By submitting you consent to receive email updates from The Biotech Catalyst Report about VolitionRx (NYSE American: VNRX). Unsubscribe anytime. No marketing churn.